How protein-based mostly COVID vaccines could trade the pandemic
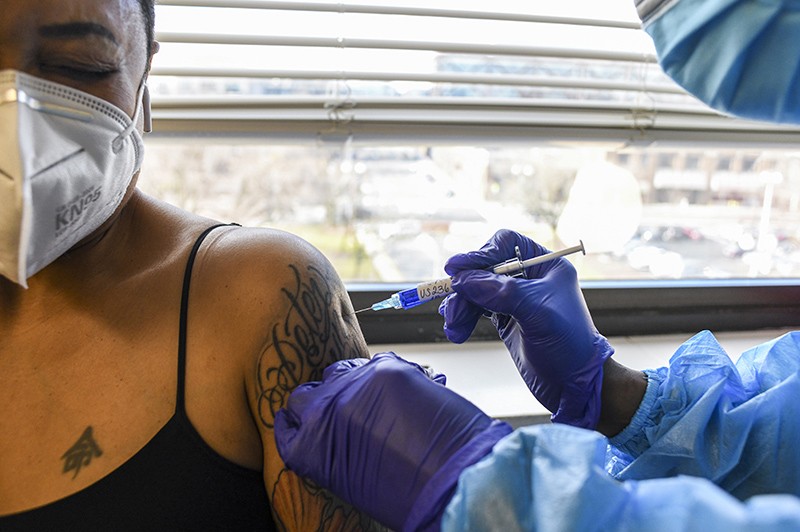

A participant in Novavax's section III trial receives a jab in early 2021.credit: Kenny Holston/The new york instances/eyevine
Pamela Sherry is desirous to turn into immunized against COVID-19. however she has delay getting a jab.
"I trust vaccines work," she says. "I desire the protection." Yet she is liable to acute immune reactions and has blood circulation problems, so she has issues concerning the photographs obtainable within the u.s., the place she lives — those in line with messenger RNA (mRNA) and viral-vector technologies. although secure for most of the population, they have got been linked to infrequent however doubtlessly severe side outcomes, including coronary heart inflammation and blood clots.
combine-and-match COVID vaccines ace the effectiveness check
So Sherry has been anticipating the menu of vaccine alternatives purchasable to her to expand. In particular, she is keeping out for a vaccine constructed from purified proteins. in contrast to the highly new technologies that the mRNA and viral-vector COVID-19 pictures are according to, protein vaccines had been used for many years to protect americans from hepatitis, shingles and other viral infections. To elicit a protecting immune response, these photographs deliver proteins, along with immunity-stimulating adjuvants, directly to someone's cells, as opposed to a fraction of genetic code that the cells should examine to synthesize the proteins themselves.
however protein vaccines don't seem to be yet in common use for COVID-19, late-stage medical-trial statistics up to now seem to be promising, demonstrating strong insurance policy with fewer side consequences than different COVID-19 photographs typically trigger.
If such a shot were attainable, "i'd go and get it right away," says Sherry, who runs a stationery company out of her home in Prosper, Texas.
Sherry's wait might quickly be over. After months of first-rate-handle setbacks and manufacturing delays, executives at biotechnology enterprise Novavax in Gaithersburg, Maryland, say they are poised to publish the enterprise's long-awaited application for his or her protein-based mostly vaccine to US drug regulators before the conclusion of the year. (On 1 November, Indonesia granted the enterprise's vaccine its first emergency authorization, and regulatory filings have already been made with government agencies in Australia, Canada, the UK, the eu Union and in different places.) in the meantime, two vaccine makers in Asia — Clover Biopharmaceuticals, primarily based in Chengdu, China, and biological E in Hyderabad, India — are in a similar fashion on course to file with a considerable number of national authorities in the coming weeks and months.

Nik Spencer/Nature
If given the go-forward, these vaccines might allay the fears of vaccine cling-outs similar to Sherry, function booster photographs, and, importantly, fill a void within the world pandemic response.
so far, fewer than 6% of individuals in low-earnings nations have been vaccinated in opposition t COVID-19. Protein-based mostly vaccines — with their comparatively cheap creation protocols and logistical benefits, together with stability at a vast latitude of temperatures — might support to slender the immunization hole between wealthy and poor nations.
"the realm needs these protein-based vaccines to reach those prone populations," says Nick Jackson, head of programmes and imaginative technologies at the Coalition for Epidemic Preparedness innovations, which has invested more than US$1 billion in five protein-based mostly COVID-19 vaccines in active building. The lion's share is going to products made with the aid of Clover, Novavax and SK bioscience in Seongnam, South Korea. "Protein vaccines are going to beckon in a brand new period of COVID-19 immunization," Jackson says.
Intrinsically sluggishFrom the earliest days of the pandemic response, researchers anticipated that protein-primarily based designs would be slower off the blocks than different vaccine applied sciences.
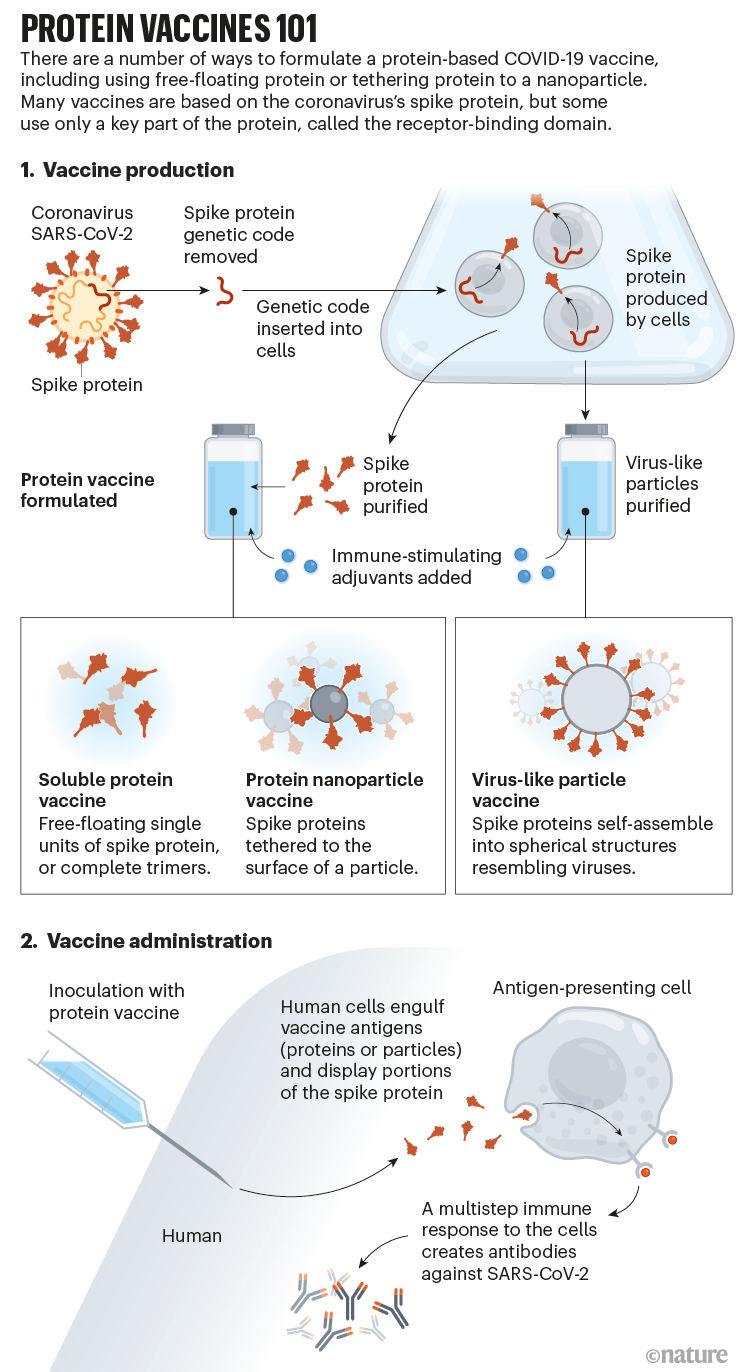
companies know the way to manufacture gobs of purified protein at scale — using genetically engineered cells from mammals, bugs or microbes — however the system includes many steps, each of which needs to be optimized for making a selected protein. "There's an intrinsic slowness," says Christian Mandl, a former trade executive who now consults on vaccine-construction issues. most of the protein-based vaccines at present in checking out have been crafted round some edition of the coronavirus SARS-CoV-2's spike protein, which helps the virus to enter cells (see 'Protein vaccines 101').

COVID vaccine boosters: probably the most crucial questions
apart from the anticipated delays, although, vaccine manufacturers made some avoidable blunders. When drug giants Sanofi and GlaxoSmithKline (GSK) teamed up on a protein vaccine assignment, for instance, onlookers anticipated scientific development to circulation with incredible haste. however the groups originally relied on misguided reagents to signify their product, leading to a dosing miscalculation. Early trial contributors acquired doses that have been about one-fifth of the planned dose.
the mistake can charge Sanofi and GSK round five months in their construction timeline, as a result of they had to repeat an exploratory study to discover the ultimate dose for late-stage testing. Their protein-based jab is now in a part III trial that kicked off in late may additionally, which contains thousands of participants in Africa, Asia and Latin the usa.
by using evaluation, significant-scale trials from Novavax and Clover have already yielded efficacy records. in keeping with a preprint published remaining month (that has no longer been peer reviewed)1, the Novavax jab provided greater than ninety% coverage in opposition t symptomatic COVID-19 in a 30,000-adult analyze completed early in the yr — before the Delta variant arrived, when only milder types of the virus have been in circulation.
Ones to watch: Vaccine makers with protein jabs in late-stage medical trialsbusiness
region
Vaccine category
mobilephone manufacturing device
organic E
Hyderabad, India
Soluble protein
Microbial cells (yeast)
Clover Biopharmaceuticals
Chengdu, China
Soluble protein
Mammalian cells (hamster ovary)
Medicago
Quebec metropolis, Canada
Virus-like particle
Plant cells (tobacco-like Nicotiana benthamiana)
Novavax
Gaithersburg, Maryland
Protein nanoparticle
Insect cells (fall armyworm)
Sanofi/GlaxoSmithKline
Paris/Brentford, UK
Soluble protein
Insect cells (fall armyworm)
SK bioscience
Seongnam, South Korea
Protein nanoparticle
Mammalian cells (human)
Clover reported a bit of lessen efficacy consequences for its protein-based mostly jab — just sixty seven% for symptomatic COVID-19 of any severity — however that quantity turned into likely deflated, since the vaccine changed into validated on populations grappling with extra virulent strains of SARS-CoV-2, together with the Delta and Mu editions. both vaccines elicited antibody stages on par with those triggered with the aid of mRNA photographs, which have emerged as one of the most most efficacious within the pandemic2,three.
The consequences exhibit that making COVID-19 vaccines the usage of proteins "isn't a substandard approach just since it took longer," says Ryan Spencer, chief executive of Dynavax applied sciences of Emeryville, California, which makes the Clover vaccine's adjuvant.

The tangled historical past of mRNA vaccines
The photographs additionally seem like safe. not one of the 50 or so protein-based mostly COVID-19 vaccines now in clinical trying out all over have elicited any fundamental aspect results. Even most of the reactions typically elicited through the mRNA or viral-vector jabs — complications, fevers, nausea and chills — have proven a ways less usual with the protein-based alternate options. as an instance, below 1% of individuals who obtained a protein-primarily based shot from Taiwan's Medigen Vaccine Biologics agency, in Taipei metropolis, developed fevers in medical reports.
"The defense profile is awfully a lot like those of influenza vaccines," says Szu-Min Hsieh, an infectious-ailment specialist at the country wide Taiwan school sanatorium in Taipei, who published section II trial consequences ultimate month4.
"That's going to enable a lot of people not to be anxious as a lot," provides Cindy gay, an infectious-ailment physician on the tuition of North Carolina school of medicine in Chapel Hill, who co-led testing of the Novavax vaccine.
Design differencesbesides the fact that one protein-based mostly vaccine succeeds — each in terms of its efficiency and in discovering a market — there's no reason to consider they all will, despite the fact.
For one thing, the form of the spike protein they install varies significantly from one product to the subsequent. Some use single proteins, others triads. Some use full-length spike protein, others simply a fragment. Some proteins are free-floating, others are packaged collectively into nanoparticles.
Why is it so tough to investigate the rare facet results of COVID vaccines?
lots of them are also manufactured the use of various kinds of mobile (see 'Ones to observe'). Novavax and Sanofi/GSK use cells from the autumn armyworm (Spodoptera frugiperda), a kind of moth, to synthesize protein; Clover and Medigen depend on hamster ovary cells, a mainstay of therapeutic antibody production within the biotechnology industry. Plus, the main candidates depend on diverse adjuvants, each and every of which prods the immune gadget in its personal manner, resulting in different types of vaccine responses.
All of this could translate into distinct efficacy and protection profiles, says Thomas Breuer, chief international fitness officer for GSK. "I may think about that you will see changes, but time and the section III trial effects will give us the best reply."
those effects have the potential to shape booster programmes in prosperous countries, where massive percentages of the population have already been vaccinated. despite the fact mRNA jabs are presently being used as boosters in lots of of those locations, tolerability concerns could power individuals to are looking for out protein-primarily based boosters once they're available.
The technology is tried and proper, and reviews have shown that a mixture-and-suit approach — in which a different COVID-19 vaccine is administered after the first — are useful at combating the sickness, notes John Mascola, director of the Vaccine analysis center at the US country wide Institute of hypersensitive reaction and Infectious ailments. "we'd deserve to see human information" confirming any such protein-primarily based booster routine is in a similar way protected and useful, Mascola says — however he and others expect that it might be. Trials evaluating the method are ongoing.
Plugging the equity holeas soon as licensed, protein pictures are also expected to swiftly handle give shortages that have plagued efforts to vaccinate lower-income countries. Novavax and Clover, as an instance, have each and every pledged to donate tons of of thousands and thousands of doses of their jabs next 12 months to COVAX, an initiative designed to distribute vaccines all over the world.
The world fitness community has also been arguing that equitable entry to COVID-19 vaccines could be achieved via native manufacturing of photographs within the world south. To obtain this, greater researchers should be seeking to primary, most economical construction techniques that manufacturers in less-prosperous international locations can without problems implement, says Christopher Love, a chemical engineer on the Massachusetts Institute of technology in Cambridge.

The combat to manufacture COVID vaccines in lessen-earnings nations
organic E is already taking expertise of 1 such equipment — yeast — to fabricate the vaccine it licensed from Baylor college of medicine in Houston, Texas. in keeping with Maria Elena Bottazzi, a Baylor virologist who helped to create the product, that makes it "doubtless the simplest and least expensive to scale" of all of the COVID-19 vaccines on or nearing the market nowadays.
within the earliest days of the COVID-19 disaster, vaccine platforms equivalent to mRNA brought the capabilities of pace, says Ralf Clemens, a vaccine-business veteran and a scientific adviser to Clover. but now that protein-based mostly vaccines are catching up, he says, they will have a lot more to present — and ultimately when it involves conserving the realm towards coronavirus infections, "I think they'll be successful."
Comments
Post a Comment